 
But if you were to take another snapshot a moment later, that same molecule might have less energy. So, if you were to take a snapshot of these particles at one moment, you might find that one molecule has a certain amount of energy. The faster particles have more energy than the slower ones. Every time they collide with each other or the walls of the container, some particles gain energy while others lose it. For example, think of gas particles bouncing around in a container. This is because molecules interact and share energy with each other. Even though they all have the same average energy, if you were to take snapshots of these molecules at different times, you'd see that their energy levels are constantly changing. To get a better grasp of entropy, imagine a group of molecules in a system that are in equilibrium. Plus, did you know that understanding entropy can help you better understand the world around you? So let's get started! What is entropy? So if you're ready to learn about the meaning of total entropy and more, keep reading! And don't worry if you're not a science whiz - we'll explain everything in simple terms that anyone can understand. We'll even show you how to calculate the entropy change of a reaction using standard entropy values. We'll explore why it's all about energy distribution, and how it relates to spontaneous reactions. In this article, we'll dive deeper into the world of entropy. 
It's like a measure of how many ways you can rearrange a bunch of puzzle pieces. But did you know that entropy is actually more complicated than that?Įntropy, or S, is all about how energy can be spread out among molecules. Since S = 0 corresponds to perfect order.If you've heard of entropy before, you might know that it's a way to measure the amount of messiness in a thermodynamic system. 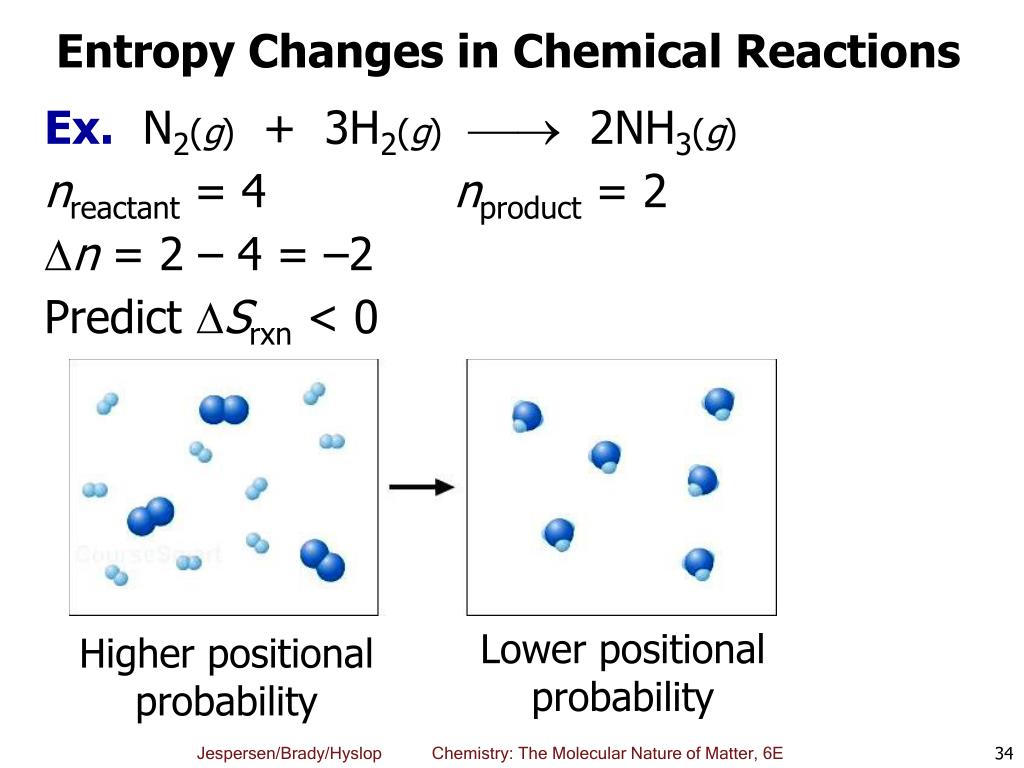
The entropy of a pure crystalline substance at absolute zero (i.e. Nonetheless, the combination of these two ideals constitutes the basis for the third law of thermodynamics: the entropy of any perfectly ordered, crystalline substance at absolute zero is zero. In practice, absolute zero is an ideal temperature that is unobtainable, and a perfect single crystal is also an ideal that cannot be achieved. Such a state of perfect order (or, conversely, zero disorder) corresponds to zero entropy. The only system that meets this criterion is a perfect crystal at a temperature of absolute zero (0 K), in which each component atom, molecule, or ion is fixed in place within a crystal lattice and exhibits no motion (ignoring quantum effects). A perfectly ordered system with only a single microstate available to it would have an entropy of zero. The greater the molecular motion of a system, the greater the number of possible microstates and the higher the entropy. These forms of motion are ways in which the molecule can store energy. due to the reduction in the degrees of freedom, the system is more ordered after the reaction). There is a reduction in the disorder of the system (i.e. 
The reaction has resulted in a loss of freedom of the atoms (O atoms).Since they are now physically bonded to the other molecule (forming a new, larger, single molecule) the O atoms have less freedom to move around.The product of this reaction (\(NO_2\)) involves the formation of a new N-O bond and the O atoms, originally in a separate \(O_2\) molecule, are now connected to the \(NO\) molecule via a new \(N-O\) bond. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
